SARS-CoV-2 (COVID19) is raging around the world, and vaccination is recognized as the most economical and effective way to control the virus epidemic. Traditional vaccine evaluation mostly uses neutralizing antibody detection methods to evaluate the effectiveness of vaccines through neutralization experiments;
Traditional methods are time-consuming and low in effificiency, usually taking 2 to 4 days to complete the evaluation, and because most of them use live viruses, It needs to be carried out in a biosafety level 3 or above laboratory, which is time-consuming and laborious, and brings great inconvenience to the evaluation of the expansion and aggregation. Therefore, there is an urgent need for a simple and rapid alternative method that is suitable for the evaluation of protective antibodies in large-scale populations.
Aehealth COVID19 Neutralization Antibody Quantitative Test Kit is used of quantitative detection of COVID19 neutralizing antibodies in human serum, plasma or whole blood. It can be used for rapid, quantitative and highly sensitive detection in vitro. clinically used in the auxiliary evaluation of the effect of the new coronavirus vaccine and evaluation of neutralizing antibodies in recovered patients after infection.
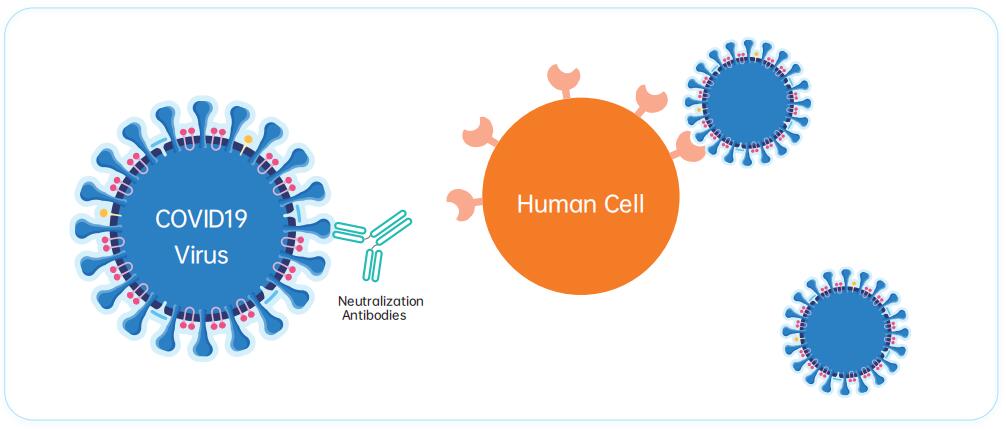
Neutralizing antibodies is effificiently stop the infection by blocking the interaction between the COVID19 virus and the host cells. Most neutralizing antibodies respond to the receptor binding domain (RBD) of the spike protein, which binds directly to the cell surface receptor ACE2. antibodies-online currently offers two neutralizing antibodies based on the clone CR3022. While most S-protein RBD binding antibodies compete for antigen binding with ACE2, the CR3022 epitope does not overlap with the ACE2-binding site.
It does thus not hinder binding of neutralizing antibodies. While CR3022 on its own exhibits only a weak neutralizing effect, it has been shown to synergize with other S-protein RBD binding antibodies to neutralize COVID19.
Easy operation
- No need professionals to be trained;
- Low sample demand, only need 50 μL;
- Compatible with multiple specimen type: Serum/Plasma/Whole blood.
High sensitivity
- Sensitivity: 98.95%;
- Specifificity: 100%.
Effificient
- Reaction time: 15 mins, Test time: 10s;
- Protable, Wide usage scenarios;
- Built-in battery, more than 200 tests without power input.
Reliable
- Verifified by 3600clinical tests, 1500 tests by infected person’s specimen,900 tests on vaccinators, 1200tests by normal people.;
- Clinical data are obtained from the vaccinators by inactivated vaccine, nucleic acid vaccine, protein vaccine, and virus infected persons, normal persons.
- Cut off value inhibition rate of 30%.
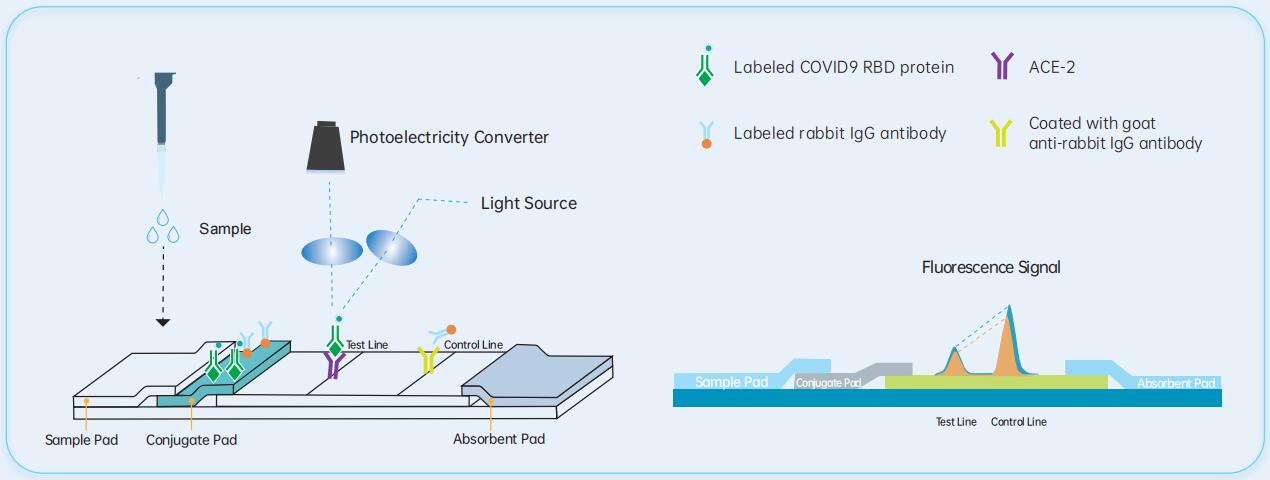
The basic principle used in this kit is immunochromatographic competition.The detection line (T-line) on the test strip is coated with angiotensin converting enzyme 2 and the control line (C-line) is coated with goat anti-rabbit IgG antibody.The conjugate pad is coated with flfluorescently labeled COVID19 RBD protein and flfluorescently labeled rabbit IgG antibody. During detection,when the sample contains the antibody to be tested, the conjugate of the test substance in the sample and the flfluorescent antigen forms an immune complex and the immune complex can no longer bind to the angiotensin-converting enzyme 2 immobilized on the nitrocellulose membrane. The flfluorescent antigen conjugate that is not bound to the antibody to be tested will bind to the angiotensin converting enzyme 2 immobilized on the nitrocellulose membrane to form a detection line (T).
- Screening test before vaccination;
- Monitoring results after vaccination;
- Risk assessment for infected people’s second infection;
- Risk assessment for normal people’s(including asymptomatic infection) possibility of infection;
- Virus resistance ability testing.

